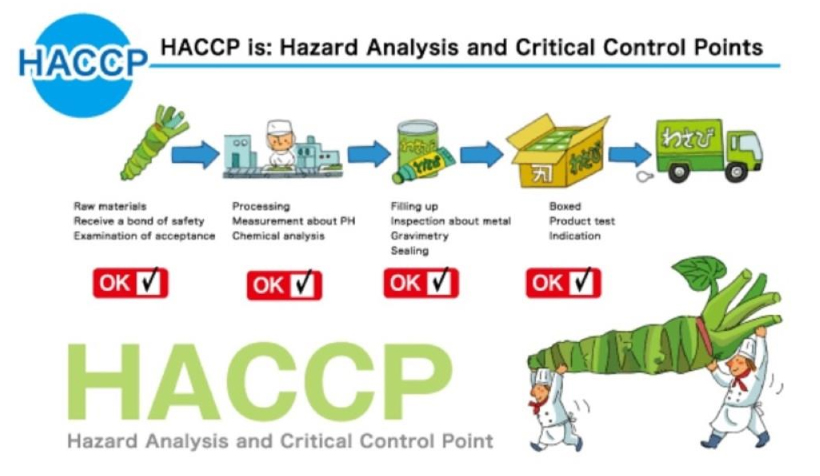
Fundamentals HACCP 食物安全重點控制
HACCP = Hazard Analysis Critical Control Point
Introduction:
- HACCP system leads to the production of microbiologically safe foods by analysing the hazards of raw materials that may appear at the processing and at the consumer end;
- The HACCP system improves product safety by anticipating and preventing health hazards before they occur;
- This HACCP concept enhances consumer confidence in the food industry while motivating developing countries to build a firm food safety control system for export;
- HACCP is the major part of requirement in ISO 22000 international standard.
History of HACCP
Pillsbury/NASA – American Space Program – 1960’s to ensure food safety of Astronauts
NASA required a “zero defect” program to guarantee safety in the foods astronauts consumed in space
It’s not a zero risk system, but is designed reduce food safety hazards to minimum
The Seven HACCP Principles
Principle 1 : Conduct Hazard Analysis
- A list of steps in the process where significant hazards occur should be prepared and their preventive measures should be described.
- Hazards are categorized into three general areas: Biological, Chemical, and Physical. For the most part, biological, which includes pathogens, is the hazard that most plans are used to prevent.
- The challenge that faces the HACCP Team is to determine what are truly significant versus insignificant hazards.
- – The team must weight both risk and severity when analyzing hazards.
Principle 2 : Determine the Critical Control Points (CCPs)
- – A critical control point is defined as a point, step or procedure at which control can be applied and a food safety hazard can be prevented, eliminated, or reduced to acceptable levels.
- – The differs from a control point, which is a less specific and important step in the process.
- – The selection of CCP is aided by the use of a CCP decision tree.
- – This decision tree is designed to allow the team to ask specific and logical questions to help determine, what is truly a critical control point versus a control point or something that could be handled under the GMPs (Good manufacturing practices) or SOPs (Standard operating procedures)
Principle 3 : Establish Critical Limit(s)
- – Critical limits must be established for preventive measures associated with each identified CCP.
– Critical limits serve as the boundaries for each CCP. Examples of critical limits are preventative measures such as temperature, pH, time, moisture level, etc.
Principle 4 : Establish CCP monitoring procedures
- Procedures must be established for using the results of monitoring to adjust the process and maintain control.
- CCP monitoring is a planned sequence of observations or measurement to assess whether a CCP is under control and to produce an accurate record for future use in verification.
- Monitoring is focused on preventing deviations (those occurrences outside the critical limits) from happening. If deviations do occur, monitoring will provide the information as to when problems occurred so that corrective action can be taken.
- Monitoring is performed best in real-time, i.e., measuring the process as it occurs rather than taking samples and sending to a laboratory.
Principle 5 : Establish Corrective Actions
- – Make practical plans for re-dressing a CCP that has gone out of “control” in advance so that actions taken are effectively, calm and planned.
Principle 6 : Establish Verification Procedures
- Verification is an on-going process that helps in ensuring that the HACCP plan is doing what it is supposed to do: prevent hazards from becoming a part of the food supply.
- Outside experts play an important role in giving input to the team as to ways to improve the plan.
Principle 7 : Establish Documentation
- The approved HACCP plan and associated records must be on file at the establishment for documenting the HACCP system.
- For the most part, this may be the only part of the HACCP plan that will be audited or reviewed by customers or regulators.
Documents include
– Temperature Chart
– Cleaning Schedules
– Pest Control Records
– Equipment Maintenance Records
– Training Records
– Delivery Checkes
– Non Conformance Reports

History of HACCP

1959
The Phillsbury Company develops concept for NASA

1971
HACCP-presented at the US National Conference on Food Protection

1972
The Pillsbury company in the United States began the application of its HACCP concept to the manufacture of its consumer food products.

1973
Pillsbury published the first comprehensive treatise on HACCP

1973
HACCP system was adopted for the Low-Acid Canned Food Regulations due to Bon Vivant Vichyssoise Soup Incidence

1980
WHO/ICMSF report on HACCP.

1983
WHO Europe recommends HACCP.

1998
HACCP becomes mandatory for large meat and poultry manufacturers

1999
HACCP becomes mandatory for small meat and poultry manufacturers

2003
FAO/WHO develop HACCP guidelines

2006
Increased worldwide use of HACCP in food safety legislation
The HACCP system has grown to become the universally accepted method for food safety assurance
Five Critical Considerations
1) The finished product and raw materials
2) The work flow (the process) no cross contamination
3) The building and equipment (design to prevent hazards not contribute to their occurence)
4) The staff, management, visitors – what hazards are they introducing
5) The procedures that are used to do instruct people in their work – do they exist, are they adequate?
Carrying out a HACCP Study
Procedure 1: Conduct a Hazard Analysis
Procedure 2: Determine the CCP's
Procedure 3: Establish Critical Limit(s)
E.g.:
-18°C
Food that is properly frozen and cooked is safe. Food that is properly handled and stored in the freezer at 0°F
(-18°C) will remain safe. While freezing does not kill most bacteria, it does stop bacteria from growing.
Procedure 4:Establish monitoring procedures
Hold such documentation and records under strict document control conditions
Procedure 5: Establish corrective actions
Procedure 6: Establish verification procedures
Procedure 7: Establish Documentation
Limitations of HACCP
HACCP requires the education of non-professional food handlers, especially in the food service industry and in homes.
The failure of the individuals to get a proper understanding of HACCP could lead to its failure.
To be effective this concept must be accepted not only by food processors but also by food inspectors and public.
It is anticipated that experts will differ as to whether a given step is a CCP and how best to monitor such steps. This has the potential of eroding the confidence of others in HACCP.
Eve after the best application of HACCP principles at the processor level, there is a need to exercise usual precautions between the purchase and consumption of the product.
Summary of HACCP
HACCP program empowers people to focus their attention on prevention rather than digging out the cause after a food borne outbreak occurs.
HACCP is not a set of seven independent principles; each must build on the other.
As with any other endeavour, all components must be working for the total plan to be effective and successful.
This system must be implemented by everyone to better assure a safer food/meat/milk/licestock product supply.
 繁體中文
繁體中文
